What are Isoxazolines?
Isoxazolines are pesticides of a chemical class introduced in the 2010s. They were launched (2013) first as veterinary products against fleas and ticks in dogs, but are also effective against numerous other external veterinary and agricultural parasites. However they seem less appropriate for use in agriculture, usage in agriculture
They can be considered as classic synthetic pesticides both by their mode of action and by their general features. They are not naturally produced molecules, but are manufactured following usual industrial chemical processes.
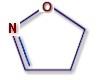
Isoxazolines are all derivatives of isoxazole (see illustration).
They have a broad spectrum of insecticidal and acaricidal activity and are effective against a number of veterinary parasites such as fleas and ticks. In laboratory studies sone of them have also shown efficacy against houseflies, mosquitoes and blowfly larvae. So far they have been introduced mainly for use in dogs and cats, and in poultry against fowl mites. Most available products (NEXGARD, NEXGARD SPECTRA, BRAVECTO, SIMPARICA, CREDELIO, etc.) are for oral administration to dogs, BRAVECTO topical solution is a topical spot-on for dogs and cats. In 2017 the first product for production animals has been introduced for use on chicken (EXZOLT)
Other isoxazolines have shown anti-microbial and anti-cancer efficacy.
Several agrochemical and pharmaceutical companies (e.g. DU PONT, BAYER, BASF, MSD ANIMAL HEALTH, NOVARTIS AH, SYNGENTA, etc.) have filed patents on various types of isoxazoline derivatives in the last years. New veterinary antiparasitics of this chemical class are likely to appear in the future. However, it seems that they are unsuitable for use in crops under field conditions and they may be used only as Animal Health products.
Click here for a general introduction to ectoparasiticides and their most important features.
Mode of action and characteristics of isoxazolines
Isoxazolines with insecticidal and tickicidal efficacy are non-competitive GABA (gamma-aminobutyric acid) receptor antagonists, much more selective for GABA receptors in insects or ticks, than for those in mammals, including humans. They bind to chloride channels in nerve and muscle cells, which blocks the transmission of neuronal signals. Affected parasites are paralyzed and die.
Isoxazolines approved for veterinary use have a systemic mode of action. Ingested or topically administered, isoxazolines are rapidly absorbed into blood and distributed throughout the whole body of the host. Blood-sucking parasites (mainly fleas and ticks) are killed during their blood meal.
But the systemic mode of action means also that for fleas and/or ticks to be killed, they have to bite the dog first and suck enough blood before the ingested active ingredient kills them. Whether killing is fast enough to prevent disease transmission is not yet known for all parasites and diseases.
Active ingredients
The isoxazolines approved for veterinary use so far are the following:
- Afoxolaner: used in dogs against fleas and some tick species (e.g. NEXGARD and NEXGARD SPECTRA introduced by MERIAL).
- Fluralaner: used in dogs and cats against fleas and some tick species (e.g. BRAVECTO oral chews and BRAVECTO topical solution introduced by MERCK ANIMAL HEALTH); and in chicken against red fowl mites (EXZOLT)
- Sarolaner: used in dogs against fleas and some tick species and certain mites (e.g. SIMPARICA introduced by ZOETIS).
- Lotilaner: used in dogs against fleas and some tick species (e.g. CREDELIO introduced by NOVARTIS).
Interestingly, both afoxolaner and fluralaner were described first by DU PONT DE NEMOURS and NISSAN, respectively, but were introduced as veterinary parasiticides by other companies (afoxolaner by MERIAL, fluralaner by MERCK ANIMAL HEALTH). This is rather unusual, since most active ingredients used in veterinary ectoparasiticides were first introduced for crop protection and only afterwards for use on animals, ivermectin being one of the very few notorious exceptions. Sarolaner and lotilaner were both discovered and developed by ZOETIS and NOVARTIS (now ELANCO), respectively, two purely animal health companies.
Safety of isoxazolines
Published information on safety and toxicity of isoxazolines is still scarce.
Target Animal Safety studies done on products approved for dogs, cats and chickens indicate that such products are well tolerated at the therapeutic dose.
In September 2018 the FDA of the USA has alerted pet owners and veterinarians about potential neurological adverse events following the use of products containing isoxazolines on dogs. In August 2021 The FDA has extended this alert to cats. Some treated animals have experienced adverse events such as muscle tremors, ataxia (lack of voluntary coordination of muscle movements), and seizures. This regards all products containing isoxazolines. Most treated animals will not show such adverse drug reactions, but some may be affected.
Additional specific information (toxicity, intoxication symptoms, adverse drug reactions, antidote, etc.) on the safety of isoxazoline active ingredients for veterinary use is available in specific articles in this site:
General information on the safety of veterinary antiparasitics is available in specific articles in this site (click to visit):
- General safety of antiparasitics for domestic animals
- General safety of antiparasitics for humans
- General safety of antiparasitics for the environment
Resistance of parasites to isoxazolines
There are so far no reports about resistance of veterinary parasites to isoxazolines, and they show no cross-resistance with other pesticides that act on GABA receptors (e.g. macrocyclic lactones and phenylpyrazoles).
Learn more about parasite resistance and how it develops.