Baylisascaris procyonis, the common raccoon roundworm, is a parasitic nematode that has raccoons as final hosts, but can infest also dogs, many other wild mammals (e.g. squirrels, mice, etc), many bird species and also humans.
Other species of the genus Baylisascaris infect several wild animals, e.g. Baylisascaris columnaris infects skunks.

Baylisascaris procyonis is not a bloodsucking worm. In its final host, the raccoon, it feeds on the intestinal content in its gut. In dogs (and humans!) it feeds on body tissues and fluids. The disease caused by this parasitic worm is called balyascariasis.
Baylisascaris procyonis is native to North America but it was brought to Europe in the early XXth century, together with raccoons that were imported for the fur industry. Since then numerous populations of wild raccoons have become established in Europe and Japan. In endemic regions up 80% of the raccoons in a population may be infected with Baylisascaris procyonis.
Baylisascaris procyonis is closely related to other roundworms of veterinary importance such as Toxocara canis, Toxocara cati, and Toxascaris leonina. All belong to a group of roundworms called ascarids (maw worms).
Baylisascaris procyonis does not affect cattle, sheep, pigs, poultry and other livestock.
Are dogs infected with Baylisascaris procyonis contagious for humans?
- NO. Because dogs only act as transport hosts of this worm species, i.e. the worms inside the dog do not shed eggs or larvae in the dog's feces that could be contagious for humans. BUT if humans accidentally ingest eggs of Baylisascaris procyonis shed by raccoons, they can indeed become infected. For additional information read the chapter on the life cycle below.
You can find additional information in this site on the general biology of parasitic worms and/or roundworms.
Final location of Baylisascaris procyonis
Predilection site of adult Baylisascaris procyonis is the small intestine, but migrating larvae in transport hosts such as dogs, many other wild mammals and also humans can be found in the abdominal cavity and in several organs (brain, lungs, eyes, heart, liver, etc.).
Anatomy of Baylisascaris procyonis
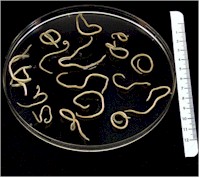
Adults of Baylisascaris procyonis have the typical slender shape of worms with thinner head and tail. They are about 15 to 20 cm long and close to 10 mm wide, whereby females are larger than males. They have a brownish to yellowish color. The worm's body is covered with a cuticle, which is flexible but rather tough.
The worms have no external signs of segmentation. They have a tubular digestive system with two openings. They also have a nervous system but no excretory organs and no circulatory system, i.e. neither a heart nor blood vessels. Males have simple chitinous spicules for attaching to the female during copulation. Each female can shed more than 100'000 eggs daily. Since infected raccoons can carry hundreds of worms, their feces can contain millions of eggs.
The eggs are almost spherical, brownish, measure about 65x80 micrometers, have a thick wall with a rough surface, and contain a single cell when shed in the raccoon's droppings.
Life cycle and biology of Baylisascaris procyonis
Baylisascaris procyonis has a direct life cycle, with raccoons as final hosts, and many mammals and birds that act as transport hosts, including dogs, squirrels, mice, birds, etc. More than 90 species of domestic and wild animals have been reported as infected by this worm. The worms do not complete development in these transport hosts, but remain encysted until a raccoon eats them or their carcasses.
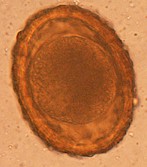
The eggs are shed with the raccoon's feces and once outside the host, mainly in the soil, L2 larvae develop inside the eggs in 2 to 4 weeks, depending on weather conditions. These eggs containing the L2 larvae are infective for dogs, other transport hosts and also humans. By suitable conditions these eggs can survive and remain infective for months and even years in the soil. They can resist dessication, freezing and exposure to temperatures higher than 50°C.
When a raccoon ingests infective eggs, they will hatch in the gut, develop to adult worms and start producing eggs. But if a dog, other transport hosts or a human person ingest such infective eggs, the larvae that hatch in the gut won't remain there, but penetrate the gut wall and start a migration (larva migrans) through the host's tissues. This migration can lead them to various organs such as the eyes (ocular larva migrans), the brain (nervous larva migrans) or other organs such as the heart, the liver, the lungs, etc, (visceral larva migrans). They do not complete development to adult worms, but remain there until a raccoon eats such an animal or its carcass. Other dog and cat roundworms behave similarly e.g. Toxocara canis, the dog roundworm, and Toxocara cati, the cat roundworm.
Once a raccoon eats such an infected intermediate host, the larval stages are liberated in the raccoon's gut, where they complete development to adult worms, mature and start producing eggs.
Harm caused by Baylisascaris procyonis, symptoms and diagnosis
Raccoons can support hundreds of worms without showing clinical symptoms.
However, infections with this worm species can be very serious for dogs and humans. Both dogs and humans become infected ingesting infective eggs that contaminate the environment (soil, vegetation, etc.). Such eggs can be abundant and particularly dangerous in and around places visited by the raccoons, which are also frequented by humans and dogs, e.g. gardens, parks, playgrounds, sandboxes, etc. This happens not only in rural environments but also in peri-urban areas that are more and more visited by raccoons.
A particular feature of migrating larvae of Baylisascaris procyonis is that almost 10% migrate to the brain of dogs or humans, where they cause a lot of damage. And in contrast with migrating larvae of other species they increase in size during their migration, which makes them more harmful. The clinical signs and the harm depend on the organs affected. Migrating larvae cause strong inflammation of the affected tissues, which causes fever and impairs the normal functioning of the affected organs. Many larvae are encysted by the host's own immune reaction and die quickly. Therefore infections with a few larvae are usually benign.
But the higher the number of migrating larvae, the higher the risk that too many survive and get into the brain. Clinical signs appear 15 to 30 days after infection. If the brain is affected, strong drowsiness, loss of coordination and sensibility, nausea and other neurological signs can occur. Fatalities due to nervous larva migrans are not uncommon. If the lungs are affected coughing, nasal discharge and other respiratory signs can be observed. Ocular larva migrans can cause light aversion, loss of vision and even blindness. Larvae migrating in the skin can cause dermatitis. Those in the gut wall can cause enteritis.
Diagnosis in raccoons is done by fecal examination for detection of eggs. In dogs and humans migrating larvae can be detected by examination of the eyes. Otherwise, diagnosis of visceral or nervoiu larva migrans is based on clinical signs and history, and ruling out other disorders. Serological assays (ELISA, immunoblotting) are available in a few research centers, but are not commercially available in many countries.
Prevention and control of Baylisascaris procyonis
Best prevention is to avoid contact with raccoons and their feces. For this reason raccoons should never be kept as pets, especially in homes with children. Wild raccoons should not be fed or otherwise be attracted to places visited by humans (e.g. leaving edible food rests outside waste bins). Raccoons use to defecate in their own "latrines" (around of trees, fallen logs, woodpiles, etc.) or inside buildings (stables, barns, attics, garages, etc.) if they can get in. Raccoon droppings (dark, tubular, strongly acidulous) and contaminated materials must be eliminated thoroughly (incinerated, buried or brought to a landfill). Use gloves and additional protective clothing (even a face mask by dry and/or windy weather) to avoid self-contamination. Afterwards wash thoroughly the hands with soap and water. Usual disinfectants may not kill the eggs. Therefore boiling water, a gas flame or power steam should be used to disinfect contaminated floors, decks or other surfaces.
All these measures are especially important in families with young children that could come in contact with raccoon's excrements. Children must be instructed to frequently clean their hands with soap before eating, to avoid contact with the pets' droppings or with dirt or soil that could be contaminated with infective eggs. These measures are obviously very advisable for adults as well. In rural and periurban areas playgrounds should be properly fenced to prevent wild raccoons to visit them.
Usual anthelmintic products (also called wormers or dewormers) containing benzimidazoles (e.g. albendazole, fenbendazole, febantel), tetrahydropyrimidines (e.g. pyrantel) or levamisole are effective against Baylisascaris procyonis infections in the raccoons. However most commercial wormers do not include this claim in their label: the responsible veterinary doctor has to determine the efficient dose tolerated by the raccoons.
There is no proven therapy against infections of dogs or humans with migrating larvae (larva migrans). There are indications that some usual wormers (e.g. albendazole) are effective against the young larvae still in the dog's gut before they start their migration. However, larvae start migration very fast, i.e. there is a very short window of opportunity for successful treatment.
There are no vaccines against Baylisascaris procyonis. To learn more about vaccines against parasites of livestock and pets click here>.
Biological control of Baylisascaris procyonis (i.e. using its natural enemies) is so far not feasible. The only option would be to eliminate the raccoons, which is neither desirable nor feasible in endemic regions.
You may be interested in an article in this site on medicinal plants against external and internal parasites.
Resistance of Baylisascaris procyonis to anthelmintics
So far there are no reports on resistance of Baylisascaris procyonisto anthelmintics when used for treating raccoons.
in raccoons, chance is very high that either the product was unsuited for the control of Baylisascaris procyonis, or it was used incorrectly.
|
Ask your veterinary doctor! If available, follow more specific national or regional recommendations for Baylisascaris procyonis control. |