Strongylus spp, also called the Large Strongyles of horses are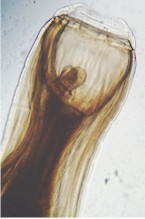 parasitic gastrointestinal roundworms that affect horses and other equids (donkeys, mules, etc.) worldwide.
parasitic gastrointestinal roundworms that affect horses and other equids (donkeys, mules, etc.) worldwide.
They are the most harmful worms of horses.
These worms do not affect cattle, sheep, goats, pigs, poultry, dogs or cats.
There are 3 different main species:
- Strongylus vulgaris, the smallest but the most harmful of all the species.
- Strongylus edentatus
- Strongylus equinus
They all are found worldwide. Depending on region and climate, up to 90% of the horses may become infected with these worms. The number of adult worms in an infected animal varies a lot.
In the last decades, intensive anthelmintic usage has led to a lower frequency of Large Strongyle infections in many places, whereas infections with Small Strongyles (Cyathostomes) has increased. This seems to be related to the fact that in most places Small Strongyles have developed anthelmintic resistance, whereas the Large Strongyles have not.
The disease caused by Strongylus worms is called strongylosis.
Are horses infected with Large Strongyles contagious for humans?
- NO: There is no evidence that these worms can infect humans.
You can find additional information in this site on the general biology of parasitic worms and/or roundworms.
Final location of Strongylus spp
Predilection site of adult Strongylus spp is the large intestine (cecum, colon). Migrating larvae can be found in several organs: particularly in blood vessels, lungs, trachea, bronchi, liver, pancreas, etc. depending on the species and the developmental stage.
Anatomy of Strongylus spp
Strongylus worms have a reddish color due to ingested blood. Strongylus vulgaris is up to 25 mm long, Strongylus edentatus up to 40 mm, and Strongylus equinus up to 50 mm. Female worms are longer than male worms. As for other roundworms, the body of these worms is covered with a cuticle, which is flexible but rather tough. The cuticle of these species shows a circular striation. The worms have a tubular digestive system with two openings.
All species have a characteristic well formed, rather spherical buccal capsule equipped with basal teeth to cut the host's tissues. They feed on blood and tissues of the organs they migrate through. These worms are so-called plug feeders, i.e. they cut out small chunks of the tissue in the organs where they stay or are migrating through.
They also have a nervous system but no excretory organs and no circulatory system, i.e. neither a heart nor blood vessels. Males have a copulatory bursa with two spicules for attaching to the female during copulation.
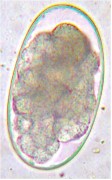
The eggs are ovoid, rather small (~45 x ~80 micrometers), thin-shelled and usually contain a 16-cell morula.
Life cycle of Strongylus spp
Strongylus worms have a direct life cycle, i.e. there are no intermediate hosts involved. Adult females lay eggs in the large intestine of the host that are shed with the feces. A single female worm can produce more than 5'000 eggs daily during several months, with peaks during the summer months in regions with moderate climate. Once in the environment the larvae hatch rather quickly and develop to L3-larvae within a few days within 2-4 days by warm and moist weather, otherwise within a few weeks, depending on temperature and humidity. Eggs do not hatch at <8°C and > 38° C. Survival of these L3 larvae is highly dependent on humidity, and dryness causes high larval mortality. By good conditions they may survive for 4 to 6 weeks in the soil.
L3-larvae are highly mobile in the soil moisture and can swim upwards in the water film that covers grass after rain or in the morning dew. This behavior favors infection during rainy weather as well as in the morning and evening hours. By dry weather and/or during intense sunshine larvae remain inside the fecal balls or swim back to the soil (negative phototaxis). In feces released in late fall egg development is often interrupted and only about 5% of these eggs may overwinter and become infective during the next spring. Survival highly depends on temperature: the colder the winter, the lower is survival.
Infection of young foals in spring is mostly due to ingestion of eggs released by their mares, which contaminate the shared pastures. It is known that egg shedding in the feces of mares increases in spring and early summer. These egg-shedding mares were already infected during the previous year and the worms they carry have overwintered in their tissues.
Infection of foals or adult horses can also occur indoors through fresh or dried hay. Hay dried on the soil is usually free of infective larvae, but larvae may survive for several weeks on rack-dried hay. Infection indoors is also possible through licking of humid walls or objects that can be contaminated with infective larvae, which are able to swim upwards in the moisture film covering the surface of such objects.
After infective L3-larvae are ingested with contaminated food or water, the subsequent life cycle and behavior is species-specific.
- Strongylus vulgaris. After being ingested, L3 larvae reach the large intestine where they penetrate in the wall of cecum and ventral colon. Here they molt to the L4-stage. About 7 days after ingestion they get into the small blood vessels of the gut wall. Once in the blood stream they migrate against the blood flow during about 2 weeks until they reach the cranial mesenteric artery. There they remain for about 4 months and complete the last molt to the pre-adult stage. Pre-adult worms migrate back to the intestine along the blood vessels, cross the gut's wall, complete development to adult worms in the lumen of the intestine, and start laying eggs that are passed in the feces. Altogether 6 to 11 months (prepatent period) after the eggs were ingested, indicating that usually there is only one generation every year.
- Strongylus edentatus. After being ingested, L3 larvae reach the large intestine where they also penetrate in the wall of cecum and ventral colon. But instead of migrating to the cranial mesenteric artery, they reach the liver through the portal vein. There they remain for about 2 months and molt to L4-larvae and cause the appearance of nodules. Afterwards they migrate to the peritoneum, where they also induce nodule formation. Subsequently they migrate further to the large intestine, where they complete the last molt, and also form nodules. These nodues eventually rupture and the adult worms reach the gut's lumen where they start laying eggs, almost a year after being ingested. The prepatent period (time between infection and first eggs shed) is 40 to 50 weeks, indicating that usually there is only one generation every year. Occasionally migrating larvae may not remain in the liver but reach such organs as the pleural cavity and the testis.
- Strongylus equinus. After being ingested, L3-larvae reach the small intestine and burrow into the submucosa of its wall, forms nodules and molt to L4-larvae within about 11 days after ingestion. Some of these L4-larvae migrate within the gut's wall during about 4 months; the rest crosses the serosa into the peritoneal cavity. There they die or penetrate into the liver where they wander through the liver tissue during about 7 weeks without forming nodules. From there they may migrate to the pancreas or other abdominal organs, molt to adult worms and after about 5 months they migrate further to the large intestine. There they reach maturity and start producing eggs, about 8 to 10 months after the eggs were ingested. The prepatent period (time between infection and first eggs shed) is 35 to 40 weeks, indicating that usually there is only one generation every year.
Harm caused by Strongylus spp, symptoms and diagnosis
Large Strongyles are the most harmful worm species to horses, particularly Strongylus vulgaris, whose larvae causes extensive damage to the mesenteric artery and its branches. Both young and adult horses can be affected. As for other parasitic roundworms, young horses are much more at risk of suffering substantial damage, not seldom fatal.
Adult worms as well as the migrating larvae are pathogenic. Damage to the gut can cause diarrhea and subsequent dehydration, colic, fever, swellings, anemia, loss of appetite (anorexia), depression, and weight loss. In severe cases gangrenous enteritis, intestinal stasis (i.e. arrest or delay of the intestinal food passage), rupture and intestinal infarct may happen, often with a fatal outcome.
Larvae in the blood vessels damage their walls, which can cause so-called verminous arteritis. Wall damage causes blood loss (hemorrhage) that favors clotting. Some clots break off and are transported elsewhere where they may obstruct the blood flow (thrombosis). If these affects the hindlimbs increasing weakness up to lameness (on one or both sides) may occur upon exercise, sometimes together with trembling and sweating. Symptoms often recede after resting.
In severe cases atrophy of the hind limb muscle may occur, as well as occlusion of the distal aorta, with acute paralysis and recumbency. Affected animals are painful and show anxiety and shock symptoms. Aneurysms, i.e. balloon-like bulges of the wall of affected blood vessels may occur as well. Such aneurysms can suddenly burst with potential fatal outcome within minutes if they affect important blood vessels.
Adults of Strongylus edentatus and Strongylus equinus can also cause significant harm to the large intestine of affected horses. However, their migratory larvae are seldom seriously damaging to the blood vessels and other organs, although they can cause liver damage and peritonitis. In contrast with this, migrating larvae are of Strongylus vulgaris are very highly pathogenic for horses with even fatal outcome, more harmful than the adult worms in the large intestine.
Adult horses that have been exposed to Strongylus spp infections develop natural immunity against these worms, and may sustain infections with almost no clinical signs. But they may remain infected and will continue shedding eggs that contaminate the pastures. This is particularly significant for the transmission of worms to young foals through infective larvae produced by their mother mares in shared pastures.
Diagnosis of is based on detection of typical eggs in the feces. However, it is almost impossible to distinguish between the three species. This usually requires coproculture to isolate the larvae, which is rather laborious. Research to develop accurate and easy-to-use diagnostic tools have not yet resulted in a commercially available test kit.
Prevention and control of Strongylus spp infections
Non-chemical prevention
An important measure to reduce the risk of infection is to avoid overstocking, because if too many animals share the same pastures, horses will rather eat grass contaminated by manure, which increases the risk of ingesting infecting larvae. Ideally each animal should be allocated 2 to 3 acres (0.8 to 1.2 hectares) of land.
If feasible, too humid pastures should be drained: the dryer the pastures, the lower the survival of infective larvae and the lower the risk of infection for the horses. Frequent manure removal is also recommended and pastures should not be fertilized with fresh manure.
Water tanks should be regularly cleaned and grazing close to them must be avoided: being wet and frequently visited they are likely to be highly contaminated with infective larvae.
To prevent infestations indoors, stable hygiene is crucial. They must be regularly cleaned, manure has to be removed daily and the bedding must be changed regularly. Humidity has to be kept as low as possible, e.g. with adequate ventilation.
Alternate grazing with livestock (cattle, sheep) that are not susceptible to Strongylus infection may be considered as well, but livestock can carry other parasites that affect horses as well.
Horses coming into a farm must be always checked for pre-existing infections (e.g. through adequate fecal examination) or treated with a broad-spectrum anthelmintic BEFORE they are allowed to share pastures and premises with other horses. In case of doubt quarantine measures must be considered.
Other preventative measures for gastrointestinal roundworms are explained in a specific article in this site (click here).
So far no true vaccine is available against Strongylus spp. To learn more about vaccines against parasites of livestock and pets click here.
Biological control of Strongylus spp (i.e. using its natural enemies) is so far not feasible. Learn more about biological control of worms.
You may be interested in an article in this site on medicinal plants against external and internal parasites.
Chemical control
Numerous broad-spectrum anthelmintics are effective against adult worms and larvae in the gut, e.g. several benzimidazoles (febantel, fenbendazole, mebendazole, oxibendazole, etc), as well as macrocyclic lactones (e.g. ivermectin, moxidectin). But not all of them are effective against migrating larvae in the tissues. Read the product label carefully to find it out. Most of them control other roundworms that may affect horses together with Strongylus spp.
A few other narrow-spectrum anthelmintics such as tetrahydropyrimidines (e.g. morantel, pyrantel) are also effective against adult worms but may not control larvae and other roundworm species that often infect livestock simultaneously with Strongylus spp.
Several commercial products contain mixtures of two or even more active ingredients of different chemical classes. This is sometimes done to extend the spectrum of activity (e.g. to ensure efficacy against roundworms and tapeworms) or to increase the chance that at least one active ingredient is effective against gastrointestinal worms that have become resistant, or to delay resistance development by those worms that are still susceptible.
Depending on the country most of these anthelmintics are available for oral administration as pastes, gels or other formulations for oral administration (e.g. tablets, drenches, feed additives). Very few are available as injectables. Many horse owners complain about the high prices of ivermectin formulations (mostly pastes or gels) for horses when compared with injectables for cattle and other livestock. The reason why ivermectin injectables are usually not used on horses is apparently that shortly after introduction, it was noticed that horses were more prone to develop severe clostridial infections at the injection site (due to contamination of the needles) and other undesired side effects than cattle or sheep. In addition, the pharmacokinetic behavior of ivermectin on horses is different than in ruminants. For these reasons oral pastes (or gels) were developed for horses that do not show such side effects.
Most wormers containing benzimidazoles (e.g. febantel, fenbendazole, oxibendazole, mebendazole, etc), tetrahydropyrimidines (e.g. morantel, pyrantel) and other classic anthelmintics kill the worms shortly after treatment and are quickly metabolized and/or excreted within a few hours or days. This means that they have a short residual effect, or no residual effect at all. As a consequence treated animals are cured from worms but do not remain protected against new infections. To ensure that they remain worm-free the animals have to be dewormed periodically, depending on the local epidemiological, ecological and climatic conditions. Exceptions to this are macrocyclic lactones (e.g. ivermectin, moxidectin) that are stored in the body fat and progressively released. They offer several weeks protection against re-infestation, depending on the dosage, the delivery form and the specific parasite. As a general rule, moxidectin achieves longer protection periods (e.g. up to ~12 weeks for suppressing egg production by the worms) than most ivermectin formulations (up to ~8 weeks).
Treatment regimes to reduce pasture contamination with infective larvae are often recommended. They are based on local climatic and ecological conditions, on farm management practices and on the approved products that vary from country to country. Ask your veterinarian for specific local recommendations.
Resistance of Strongylus spp to anthelmintics
So far there are no reports on significant problems due to Strongylus spp resistance to most commonly used anthelmintics.
This means that if an anthelmintic fails to achieve the expected efficacy against these worms, chance is significant that it is not due to resistance but to incorrect use, which is the most frequent cause of product failure. Or the product used was not adequate for the control of Strongylus spp.
However, Large Strongyles often infect horses together with Small Strongyles (mixed infections), which on their turn have developed resistance to most common anthelmintics in many places.
Learn more about parasite resistance and how it develops.
|
Ask your veterinary doctor! If available, follow more specific national or regional recommendations for Strongylus spp control. |