Thiametoxam is an antiparasitic active ingredient used in livestock premises mainly against flies. It is also used against agricultural and household pests. It belongs to the chemical class of the neonicotinoids.
Common name: THIAMETHOXAM
Type: Pesticide
Chemical class: neonicotinoid
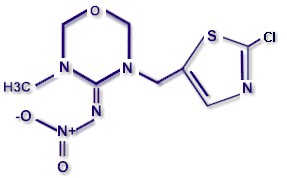
CHEMICAL STRUCTURE
EFFICACY AGAINST PARASITES
Type of action: Contact, non-systemic insecticide
Main veterinary parasites controlled: houseflies, filth & nuisance flies
Efficacy against a specific parasite depends on the delivery form and on the dose administered. National regulatory authorities determine whether a product is approved for a given indication, i.e. use on a particular host at a specific dose and against a specific parasite. Check the labels of the products available in your country.
Click here for general information on features and characteristics of PARASITICIDES.
SAFETY
Oral LD50, rat, acute*: 1563 mg/kg
Dermal LD50, rat, acute*: >2000 mg/kg
* These values refer to the active ingredient. Toxicity has to be determined for each formulation as well. Formulations are usually significantly less toxic than the active ingredients.
MRL (maximum residue limit): Not applicable: not approved for livestock
Withholding periods for meat, milk, eggs: Not applicable: not approved for livestock
Recently (April 2013) the EU has prohibited for two years the use of several nicotinoids on various crops due to suspected detrimental effects on bee colonies. This should have no influence on veterinary uses of neonicotinoids.
General safety information for antiparasitics is available in specific articles in this site (click to visit):
- General safety of antiparasitics for domestic animals
- General safety of antiparasitics for humans
- General safety of antiparasitics for the environment
|
WARNING Never use agricultural or hygiene products with this or any other active ingredient on livestock or pets, even if there are veterinary products with this same active ingredient approved for use on animals. The formulations for agricultural or hygiene use are different and may be toxic for livestock or pets. It is obvious that veterinary products are not intended for and should never be used on humans!!! |
MARKETING & USAGE
Decade of introduction: 1990
Introduced by: NOVARTIS (→ELANCO)
Some original brands: AGITA
Patent: Expired (ex SYNGENTA; particular formulations may be still patent-protected)
Use on LIVESTOCK: Not on animal. Only off-animal use in livestock and agricultural premises premises.
Use on HORSES: NO
Use on DOGS and CATS: No
Main delivery forms:
Use in human medicine: No
Use in public/domestic hygiene: No
Use in agriculture: Yes
Generics available: Yes
PARASITE RESISTANCE
On livestock: Yes, reported on houseflies (Musca domestica) in Denmark (resistance factor 6 to 76), even before introduction of neonicotinoids in the Danish market!
SPECIFIC FEATURES
Thiamethoxam is a neonicotinoid used only off-animal for the control of houseflies and other nuisance flies in livestock operations (poultry houses, stables, piggeries, etc.). It is also used massively in agriculture.
It acts mainly as an oral insecticide for flies, i.e. it does not control bloodsucking flies or any other insects.
It can be used as a scatter bait, or as a paint-on for treating the surfaces where the flies use to rest.
Thiamethoxam is NOVARTIS' succesor of azamethiphos (SNIP, ALFACRON), which was phased out by the company in the 2000's when it decided for strategic and image reasons to get rid of all organophosphates in its product range.
Mechanism of action of thiamethoxam
As all neonicotinoids thiamethoxam is an agonists of the nicotinic acetylcholine receptors. It takes the place of the normal neurotransmitter acetylcholine in the receptors, which cannot be deactivated by acetylcholinesterase and remains irreversibly blocked. This leads to an over stimulation of the nerve cells, to paralysis and to death of the affected insect.
These receptors are found in the central and peripheral nervous system of mammals, but only in the central nervous systems of insects. Neonicotinoids bind much more strongly to insect receptors than to mammal receptors, which makes them relatively safe for domestic animals and humans.
Click here to view the list of all technical summaries of antiparasitic active ingredients in this site.