Niclosamide is an antiparasitic active ingredient used in veterinary and human medicine. It is used mainly against tapeworms of dogs, cats, horses and livestock. It is also used in agriculture against snails. It belongs to the chemical class of the salicylanilides.
Common name: NICLOSAMIDE
Type: veterinary medecine
Chemical class: salicylanilide
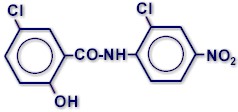
CHEMICAL STRUCTURE
EFFICACY AGAINST PARASITES
Type of action: Taenicide anthelmintic, endoparasiticide.
Main veterinary parasites controlled: tapeworms (=cestodes), rumen flukes (Paramphistomum spp)
Efficacy against a specific parasite depends on the delivery form and on the dose administered.
Click here for general information on features and characteristics of PARASITICIDES.
DOSING
Click here to view the article in this site with the most common dosing recommendations for niclosamide used in domestic animals.
SAFETY
Oral LD50, rat, acute*: >5000 mg/kg
Dermal LD50, rat, acute*: >2000 mg/kg
* These values refer to the active ingredient. Toxicity has to be determined for each formulation as well. Formulations are usually significantly less toxic than the active ingredients.
MRL (maximum residue limit) established for either beef, mutton pork or chicken meat*:
- CODEX: No
- EU: No
- USA: No
- AUS: No
* This information is an indicator of the acceptance of an active ingredient by the most influential regulatory bodies for use on livestock.
Withholding periods for meat, milk, eggs, etc. depend on delivery form, dose and national regulations. Check the product label in your country.
Learn more about niclosamide safety (poisoning, intoxication, overdose, antidote, symptoms, etc.).
General safety information for antiparasitics is available in specific articles in this site (click to visit):
- General safety of antiparasitics for domestic animals
- General safety of antiparasitics for humans
- General safety of antiparasitics for the environment
|
WARNING Never use agricultural or hygiene products with this or any other active ingredient on livestock or pets, even if there are veterinary products with this same active ingredient approved for use on animals. The formulations for agricultural or hygiene use are different and may be toxic for livestock or pets. It is obvious that veterinary products are not intended for and should never be used on humans!!! |
MARKETING & USAGE
Decade of introduction: 1950
Introduced by: BAYER
Some original brands: MANSONIL, LINTEX, YOMESAN
Patent: Expired (particular formulations may be still patent-protected)
Use in LIVESTOCK: Yes, very scarce
Use in HORSES: NO
Use in DOGS and CATS: Yes, very scarce
Main delivery forms:
Use in human medicine: Yes
Use in public/domestic hygiene: Yes, against snails
Use in agriculture: Yes, against snails
Generics available: Yes, a few
PARASITE RESISTANCE
In pets: No
In livestock: No
Learn more about parasite resistance and how it develops.
SPECIFIC FEATURES
Niclosamide is a veteran salicylanilide anthelmintic, nowadays very scarcely used in pets and livestock, because it has been replaced by more effective compounds. It is available in a few countries in the form of drenches, tablets, pills, etc. and feed additives.
Due to its narrow spectrum of activity it is mostly used in combination with a broad-spectrum nematicide (e.g. benzimidazoles, levamisole, etc.).
Efficacy of niclosamide
Niclosamide is a good taenicide (= tapeworm killer) effective against several parasitic tapeworms of livestock and pets (e.g. Taenia spp, Moniezia spp) and also against rumen flukes (Paramphistomum spp) and blood flukes (Schistosoma spp). This is in contrast with most other salicylanilides, which are mainly flukicides and not at all taenicides.
Niclosamide is also used against aquatic snails that are vectors of human schistosomiasis (also known as bilharzia, bilharziosis or snail fever), as well as a human medicine against various tapeworm infections.
It has no efficacy whatsoever against roundworms (nematodes) and other important parasitic flukes such as liver flukes (Fasciola hepatica), nor against external parasites.
Niclosamide has no residual effect. This means that a single administration will kill the parasites present in the host at the time of treatment, but will not protect the host against re-infestations.
Pharmacokinetics of niclosamide
After oral administration niclosamide is only marginally absorbed into the bloodstream. Absorbed niclosamide is quickly metabolized and excreted. Unabsorbed niclosamide remains vastly unchanged in the digestive tract and is quickly excreted through feces. Neither the parent molecule nor its metabolites are detected in blood or urine.
This low absorption allows a high safety margin for use on animals.
Mechanism of action of niclosamide
The molecular mode of action of salicylanilide, including niclosamide, is not completely elucidated. They all are uncouplers of the oxidative phosphorylation in the cell mitochondria, which disturbs the production of ATP, the cellular "fuel". This impairs the parasites motility and probably other processes as well.
Niclosamide acts on the tapeworms also through inhibition of glucose absorption.
Click here to view the list of all technical summaries of antiparasitic active ingredients in this site.